 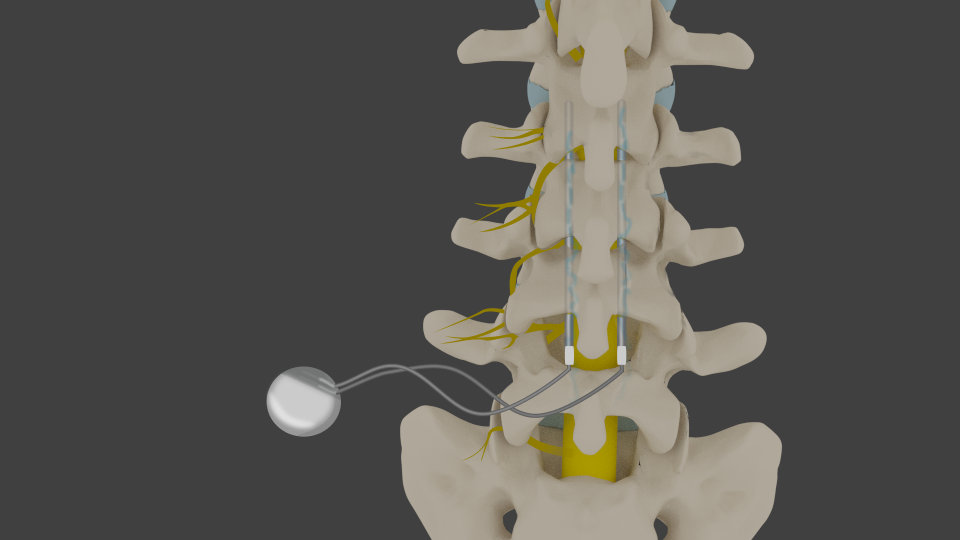 
This system is contraindicated for patients who are unable to operate the system or who have failed to receive effective pain relief during trial stimulation. Indications For UseĪbbott Medical spinal cord stimulation (SCS) systems are indicated as an aid in the management of chronic, intractable pain of the trunk and/or limbs, including unilateral or bilateral pain associated with the following: failed back surgery syndrome, nonsurgical back pain (without prior surgery and not a candidate for back surgery), and diabetic peripheral neuropathy of the lower extremities. The system is intended to be used with leads and associated extensions that are compatible with the system. This neurostimulation system is designed to deliver low-intensity electrical impulses to nerve structures. 
All use the clinically proven low-dose BurstDR stimulation therapy.Spinal Column Stimulation (SCS) Systems Intended Use iPhone integration: Abbott’s patient-focused mobile app supports real-time battery life and charging status updates of the Eterna SCS system on a personal or Abbott-provided Apple device.Ībbott’s portfolio of neuromodulation devices also includes Proclaim™ XR, the recharge-free SCS system, and Proclaim Plus featuring FlexBurst360™, the SCS system that offers pain coverage across up to six areas of the trunk and/or limbs and enables programming that can be adjusted as a person’s therapeutic needs evolve.Upgradable platform: As Abbott continues to make advancements in SCS therapy, the Eterna SCS system can adapt to future innovations without replacing the implant.MRI capabilities: Eterna uses Abbott’s TotalScan™ MRI technology, which allows for full-body MRI scans – a critical need of chronic pain patients who require accessibility to improved diagnostics and healthcare.Lowest recharging requirements: Paired with Abbott’s new Xtend™ energy technology, Eterna optimizes the patient charging experience, requiring as few as five recharges per year under standard use from a wireless charger.BurstDR stimulation is preferred to traditional “tingling” tonic stimulation by 87% of patients. Clinically proven therapy: The Eterna SCS system features Abbott’s proprietary BurstDR stimulation, which mimics natural firing patterns found in the brain to deliver superior pain relief.Smallest device: Designed with daily comfort in mind6 the Eterna SCS system is the smallest implantable, rechargeable spinal cord stimulator on the market.With patient needs front of mind, Abbott created Eterna to be recharged less than five times a year under normal use, making it the lowest recharge burden platform on the market. The studies found that people wanted a smaller implant for comfort while reducing the need to charge the device daily or weekly. Decem– Abbott announced the FDA approval of the company’s Eterna™ spinal cord stimulation (SCS) system – the smallest implantable, rechargeable spinal cord stimulator currently available on the market for the treatment of chronic pain. Eterna SCS utilizes Abbott’s proprietary low-dose BurstDR™ stimulation, the only SCS waveform technology with the highest level of clinical evidence (1A evidence), proven to reduce pain 23% more than traditional waveform technology approaches.Ībbott developed Eterna based on extensive studies with patients, physicians and caregivers to understand the unmet needs of people living with chronic pain. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
